More Information
Submitted: March 31, 2026 | Accepted: April 07, 2026 | Published: April 09, 2026
Citation: Elafari MA, Ayoub M, Bibat MA, Slaoui A, Karmouni T, Koutani A, et al. Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical Recovery. J Clin Med Exp Images. 2026; 10(1): 032-038. Available from:
https://dx.doi.org/10.29328/journal.jcmei.1001046.
DOI: 10.29328/journal.jcmei.1001046
Copyright license: © 2026 Elafari MA, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Renal malakoplakia; Michaelis-gutmann bodies; Diabetes mellitus; Escherichia coli; Nephrectomy; Granulomatous inflammation; Von hansemann cells; DMSA scintigraphy
Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical Recovery
Mohammed Amine Elafari* , Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tarik Karmouni, Abdelatif Koutani and Khalid Elkhader
, Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tarik Karmouni, Abdelatif Koutani and Khalid Elkhader
Urology B Department, Ibn Sina Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco
*Corresponding author: Mohammed Amine Elafari, Urology B Department, Ibn Sina Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco, Email: [email protected]
Background: Renal malakoplakia is a rare chronic granulomatous inflammatory disorder characterized by defective macrophage function. It typically occurs in immunocompromised patients with recurrent urinary tract infections. We present a case of renal malakoplakia in a diabetic patient who progressed to nephrectomy despite initial conservative management.
Case presentation: A 57-year-old female patient with a medical history of insulin-dependent type 2 diabetes mellitus was admitted to the hospital with symptoms including fever, left flank pain, and dysuria. A physical examination revealed a tender left lumbar mass. Laboratory investigations revealed a leukocytosis (16,500/mm³), elevated C-reactive protein (142 mg/L), and preserved renal function. A urine culture revealed the presence of multidrug-resistant Escherichia coli (>106CFU/mL). A subsequent Computed Tomography (CT) scan revealed an enlarged left kidney with a 9 × 6 cm multiloculated subcapsular collection, causing significant parenchymal compression, along with two non-obstructive inferior pole calculi. The initial management strategy encompassed ultrasound-guided percutaneous drainage and targeted antibiotic therapy, with the latter being contingent upon bacterial sensitivities. Notwithstanding the patient’s positive clinical recovery, Technetium-99m Dimercaptosuccinic Acid ((99m)Tc-DMSA) renal scintigraphy performed four weeks after the episode revealed a non-functional left kidney, exhibiting a 15% differential function. Following a multidisciplinary discussion, a total left nephrectomy was performed. A histopathological examination revealed extensive replacement of renal parenchyma by polymorphous inflammatory infiltrate with pathognomonic Michaelis-Gutmann bodies. These bodies are spherical, basophilic, perinuclear inclusions that demonstrate strong positivity for Periodic Acid-Schiff and Perls stains. The postoperative course was complicated by self-limited lymphorrhage. At the 3-month follow-up, the patient reported complete resolution of symptoms and remains under nephrological surveillance.
Conclusion: This case underscores the diagnostic challenges posed by renal malakoplakia, a condition that can present with a wide spectrum of mimics, including infectious and neoplastic processes. Early diagnosis and prolonged antibiotic therapy with agents capable of intracellular penetration may preserve renal function; however, nephrectomy remains necessary when irreversible parenchymal damage has occurred. Diabetes mellitus has been identified as a significant risk factor for malakoplakia development through impaired leukocyte function.
Malakoplakia, a term derived from the Greek words “malakos” (soft) and “plakos” (plaque), is an uncommon chronic granulomatous inflammatory disorder first described by Michaelis and Gutmann in 1902 [1]. While the condition primarily affects the genitourinary tract, renal parenchymal involvement is a particularly rare manifestation that predominantly affects middle-aged women, with a female-to-male ratio of 3:1 [2].
As a diagnostic challenge, its primary teaching point lies in its ability to mimic neoplastic processes while causing silent, irreversible parenchymal destruction despite apparent clinical recovery. Although global renal function often remains stable, as reflected by serum creatinine levels, due to contralateral compensation, the affected kidney may lose its functional viability entirely. This underscores the necessity for a shift from purely clinical monitoring to early histopathological and functional evaluation to preserve renal health.
The pathogenesis of malakoplakia is characterized by defective macrophage bactericidal function, resulting in incomplete digestion of phagocytosed bacteria, most commonly Escherichia coli [3]. This phenomenon results in the accumulation of partially degraded bacterial remnants within macrophages, leading to the formation of characteristic intracytoplasmic inclusions known as Michaelis-Gutmann bodies. These distinctive structures are indicative of calcospherites, which are composed of calcium and iron deposits encircling incompletely digested bacterial fragments [4].
Risk factors for malakoplakia include immunocompromised states such as organ transplantation, malignancy, autoimmune disorders, and diabetes mellitus [5,6]. The association with diabetes mellitus is particularly salient, as hyperglycemia impairs leukocyte function, creating a permissive environment for persistent bacterial infection and subsequent malakoplakia development [7].
Renal parenchymal malakoplakia has historically been associated with a poor prognosis, characterized by high mortality rates. Nevertheless, significant advancements in diagnostic imaging, early tissue diagnosis, and antimicrobial therapy (particularly fluoroquinolones and other agents with intracellular penetration) have resulted in substantial improvements in patient outcomes [8]. However, cases exhibiting significant parenchymal destruction may necessitate nephrectomy [9].
We hereby present a case of renal malakoplakia in a diabetic patient, which illustrates the clinical presentation, diagnostic approach, and therapeutic decision-making in this rare entity.
A 57-year-old female patient with a long-standing medical history of insulin-dependent type 2 diabetes mellitus was admitted to our institution with symptoms including fever, left flank pain, and persistent dysuria. The patient reported chronic, recurrent lumbar discomfort for which she had previously neglected to seek medical intervention. The patient denied symptoms of hematuria, weight loss, or constitutional symptoms beyond fever.
A physical examination of the patient revealed that their hemodynamic status was stable, with normal vital signs apart from fever. The general condition appeared to be satisfactory. A thorough abdominal examination revealed a palpable left lumbar mass on deep palpation, accompanied by a positive renal contact sign. A thorough examination of the pelvis revealed no indications of gynecological pathology.
Laboratory investigations confirmed a systemic inflammatory response. A complete blood count revealed a leukocytosis of 16,500/mm³, with a predominance of 85% neutrophil. C-reactive protein levels were found to be significantly elevated at 142 mg/L. Renal function was maintained, as indicated by a serum creatinine level of 9 mg/L and an estimated glomerular filtration rate of 78 mL/min/1.73m².
A urinalysis revealed the presence of pyuria and bacteriuria. A urine culture revealed the presence of multidrug-resistant Escherichia coli (E. coli) (>10⁶CFU/mL). Consequently, specialized testing for Mycobacterium tuberculosis yielded a negative result, thereby excluding the diagnosis of tuberculous pyelonephritis from the differential diagnosis.
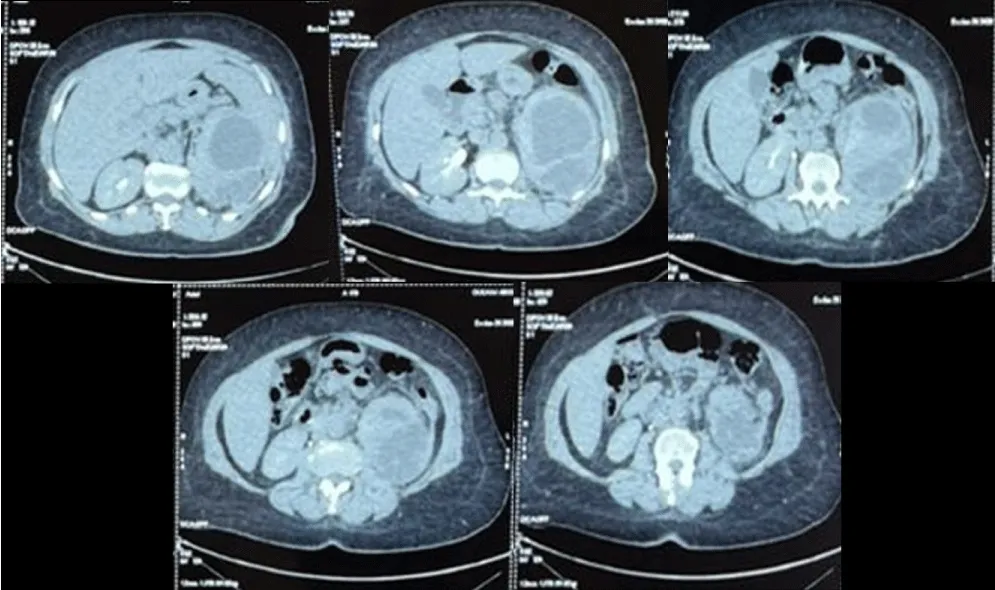
A subsequent Computed Tomography (CT) imaging of the abdomen and pelvis revealed an enlarged left kidney with a 9 × 6 cm multiloculated subcapsular collection characterized by a thick, enhancing wall with internal septations, causing a ‘mass effect’ that compressed the collecting system without causing downstream ureteral obstruction. Two non-obstructive calculi were identified in the inferior pole, measuring 9 millimeters and 3 millimeters, respectively, with attenuation of 274 Hounsfield Units (HU), consistent with calcium-containing stones. It is noteworthy that there was an absence of evidence indicative of collecting system dilatation or ureteral obstruction (Figure 1).
Figure 1: Axial contrast-enhanced CT scan of the abdomen. The images reveal an enlarged left kidney with a 9 × 6 cm multiloculated subcapsular collection. This collection causes significant compression of the renal parenchyma and lamination of the calyceal groups. No evidence of collecting system dilatation or ureteral obstruction is noted.
The initial management strategy combined ultrasound-guided percutaneous drainage of the subcapsular collection with empiric intravenous antibiotic therapy. Following the identification of multidrug-resistant E. coli (which demonstrated resistance to amoxicillin-clavulanate, ciprofloxacin, and trimethoprim-sulfamethoxazole), antimicrobial treatment was tailored based on sensitivity patterns. The therapy was narrowed to intravenous cefepime (2g every 12 hours), as the isolate remained sensitive to both cefepime (MIC ≤ 1 µg/mL) and amikacin. Consequently, the patient showed a favorable clinical response characterized by the resolution of fever and flank pain, accompanied by a marked decrease in inflammatory markers.
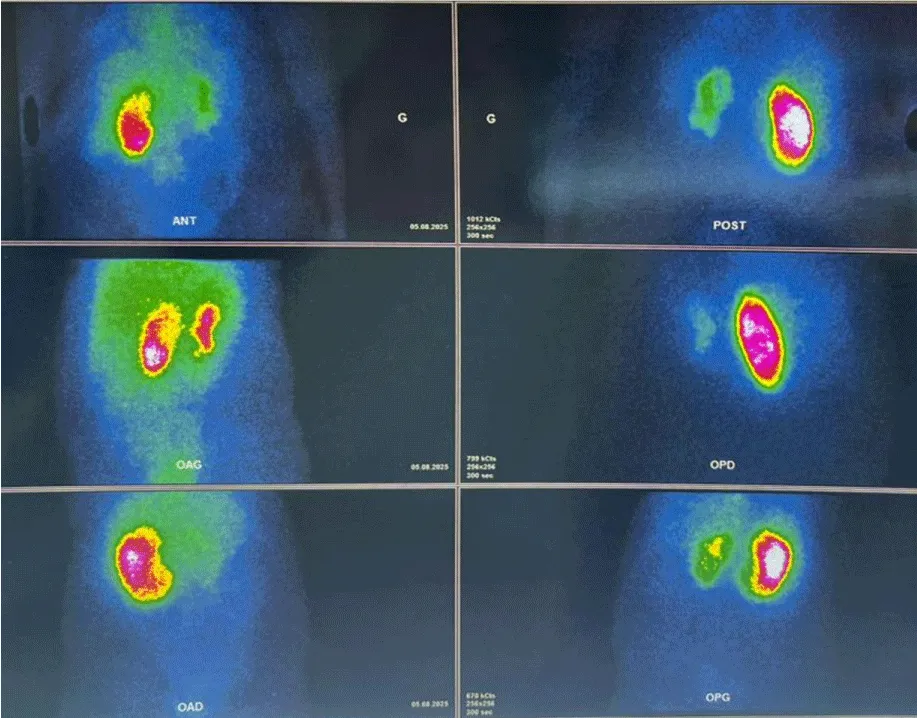
Despite the evident clinical and biological recovery, concerns regarding long-term renal function persisted. A Technetium-99m dimercaptosuccinic acid (99mTc-DMSA) renal scintigraphy was performed four weeks after the episode to assess differential renal function. The study revealed a non-functional left kidney, which contributed a mere 15% of total renal function, indicating severe irreversible parenchymal damage (Figure 2).
Figure 2: (99m)Tc-DMSA renal scintigraphy. Functional imaging was performed four weeks after the initial acute episode. The scan demonstrates a severely hypo-functional left kidney, contributing only 15% to the total differential renal function. These findings indicate irreversible parenchymal damage despite the patient's clinical and biological recovery.
Following a comprehensive review, the case was presented to a multidisciplinary team comprising nephrologists, urologists, infectious disease specialists, and radiologists. Given the clinical complexity, the decision was made to proceed with a total left nephrectomy. This surgical intervention was justified by three critical factors: the severe and irreversible loss of renal function (demonstrated as <20% differential function on scintigraphy), the persistent risk of recurrent sepsis in an immunocompromised diabetic host, and the diagnostic necessity to definitively exclude an underlying malignancy.
A total laparoscopic left nephrectomy was performed. The intraoperative course was uneventful, with no significant technical difficulties encountered. Postoperative complications manifested briefly in the form of lymphorrhage. This issue was addressed through a conservative management approach, which included bed rest and compression. Following these interventions, lymphorrhage resolved without the need for further treatment. The patient did not require surgical re-intervention or blood transfusion.
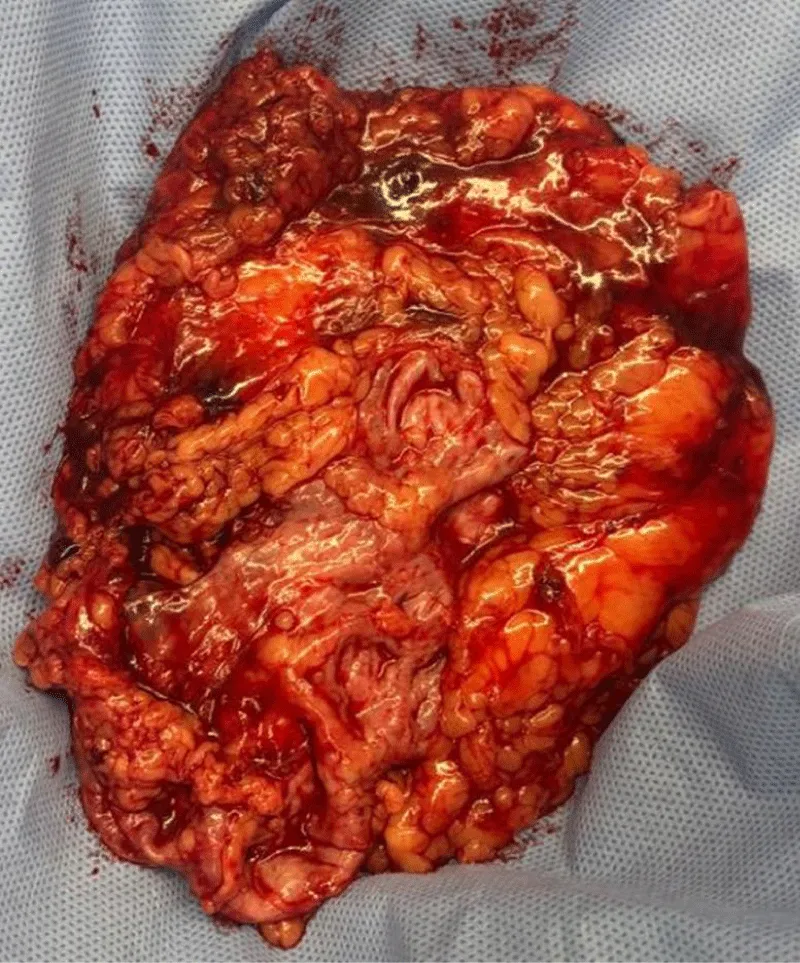
The histopathological examination of the surgical specimen yielded the definitive diagnosis. Gross examination revealed an enlarged kidney with extensive parenchymal destruction and replacement by inflammatory tissue. The subcapsular collection was found to contain purulent material (Figure 3).
Figure 3: Gross examination of the total left nephrectomy specimen. The surgical specimen shows an enlarged kidney with extensive destruction of the normal architecture. The parenchyma is largely replaced by yellowish-tan inflammatory tissue, and the subcapsular space contains residual purulent material.
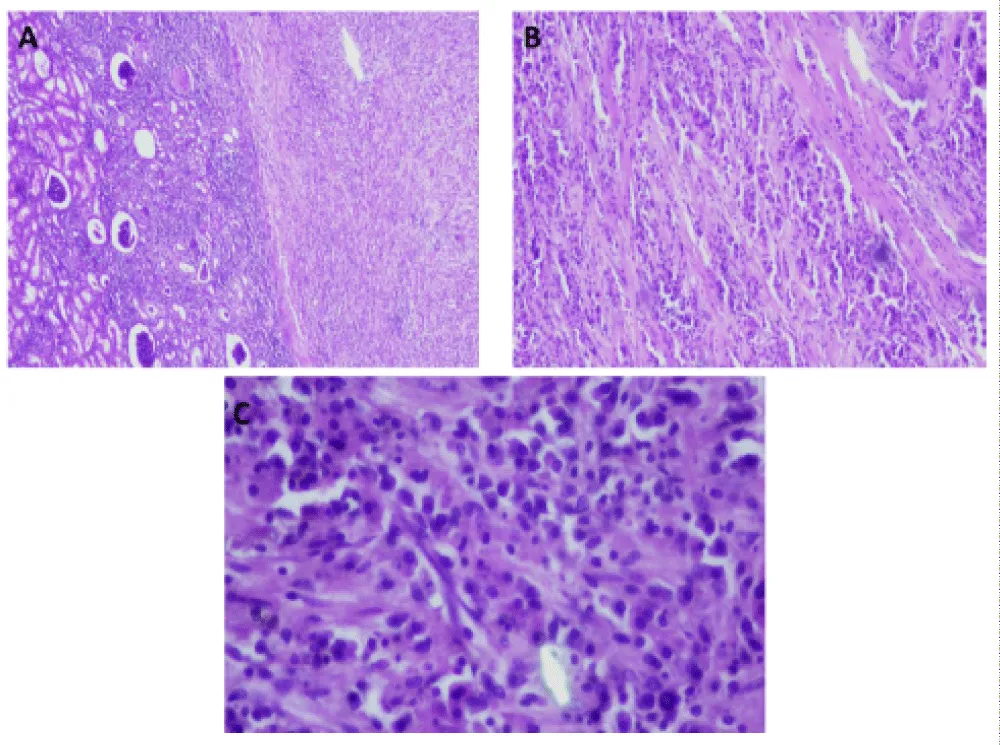
Microscopic examination revealed significant replacement of normal renal parenchyma by a diffuse and nodular polymorphous inflammatory infiltrate. The infiltrate was predominantly composed of lymphocytes and histiocytes. The histiocytes manifested an abundance of eosinophilic cytoplasm, a hallmark feature of von Hansemann cells.
The diagnosis was confirmed by the presence of Michaelis-Gutmann bodies, which are spherical, basophilic, perinuclear inclusions with a characteristic “target-like” or “owl’s eye” appearance within the histiocytic cytoplasm. These inclusions exhibited a strong positivity for Periodic Acid-Schiff (PAS) stain, thereby indicating their glycoprotein content. Furthermore, the Perls Prussian blue stain for iron exhibited a strong positive reaction, thereby confirming the presence of iron deposits within the calcospherites (Figure 4).
Figure 4: Histopathological examination of the renal parenchyma. (A) Low-power view (H&E): Extensive infiltration by a sheet of histiocytic cells mingled with polymorphonuclear neutrophils and plasma cells (left), replacing the normal renal architecture (right). (B) Medium-power view (H&E): High concentration of von Hansemann cells, identified as large histiocytes with abundant, granular eosinophilic cytoplasm. (C) High-power view (H&E): Detailed view of histiocytes containing eosinophilic intracytoplasmic inclusions (von Hansemann cells) and pathognomonic Michaelis–Gutmann bodies. The latter appear as small, basophilic, rounded intracytoplasmic inclusions.
Notably, the absence of granulomatous inflammation, indicative of mycobacterial or fungal infection, was observed. No malignancies were identified, precluding the possibility of renal cell carcinoma or other neoplastic processes from the differential diagnosis.
At the three-month postoperative follow-up, the patient reported a complete resolution of symptoms, including flank pain and dysuria. After the initial infection, the patient did not manifest any additional episodes of urinary tract infection. The patient is undergoing regular nephrological surveillance to optimize management of her solitary right kidney and ensure adequate glycemic control to prevent further complications. Renal function has remained stable, with serum creatinine levels measuring 10 mg/L (Table 1).
| Discussion: Clinical timeline, interventions, and diagnostic investigations. | ||
| Timing | Clinical Event / Intervention | Key Investigations & Results |
| Day 1 | Admission: Fever, left flank pain, and dysuria. | WBC: 16,500/mm³; CRP: 142 mg/L. CT scan: 9 × 6 cm multiloculated subcapsular collection. |
| Day 2 | Initial Management: Ultrasound-guided percutaneous drainage and empiric IV antibiotics. | Drainage of purulent material. |
| Day 4 | Targeted Therapy: Antibiotic narrowing based on sensitivity patterns. | Urine Culture: MDR E. coli (>10⁶ CFU/mL), sensitive to Cefepime (MIC ≤ 1 µg/mL). |
| Week 4 | Functional Assessment: Clinical and biological recovery noted. | (99m)Tc-DMSA Scintigraphy: Non-functional left kidney (15% differential function). |
| Week 6 | Surgical Intervention: Multidisciplinary team decision for total laparoscopic left nephrectomy. | Histopathology: Presence of von Hansemann cells and pathognomonic Michaelis-Gutmann bodies. |
| Month 3 | Follow-up: Complete resolution of symptoms. | Stable renal function (Creatinine: 10 mg/L) under nephrological surveillance. |
This case illustrates several important aspects of renal malakoplakia, a rare but clinically significant entity that poses diagnostic and therapeutic challenges.
The pathogenesis of renal malakoplakia is centered on defective macrophage bactericidal function, specifically characterized by impaired phagolysosomal digestion. The prevailing theory posits that affected macrophages exhibit reduced cyclic Guanosine Monophosphate (cGMP) levels and impaired microtubule assembly, leading to incomplete bacterial killing. This phenomenon enables bacterial antigens, particularly those derived from E. coli, to persist within the cells and induce a persistent inflammatory response [2].
In our case, the presence of persistent Lipopolysaccharide (LPS) from incompletely digested E. coli fragments likely triggered prolonged inflammatory cascades. Furthermore, recent insights suggest that the Sirtuin 1 (SIRT1) pathway, which regulates macrophage inflammatory responses, may hold significant clinical relevance. In a diabetic host like our patient, hyperglycemia-induced SIRT1 downregulation could theoretically exacerbate macrophage dysfunction by preventing the transition from a pro-inflammatory to a reparative phenotype, thereby accelerating irreversible parenchymal fibrosis.
This molecular environment, combined with the patient’s insulin-dependent diabetes mellitus, a well-recognized risk factor, creates a permissive setting for malakoplakia development. Diabetes impairs multiple aspects of immune function, including neutrophil chemotaxis, phagocytosis, and intracellular killing, while hyperglycemia fosters a conducive environment for bacterial proliferation [5-7].
The hallmark of this disorder is the formation of pathognomonic Michaelis-Gutmann bodies. These structures develop when partially digested bacterial fragments accumulate within phagolysosomes, creating a focal point for calcium and iron deposition [4]. The resulting calcospherites manifest as concentrically laminated, basophilic inclusions that serve as the definitive diagnostic marker for the condition.
Renal Parenchymal Malakoplakia (RPM) is a rare condition that manifests with nonspecific symptoms, including fever, flank pain, and recurrent urinary tract infections. These symptoms often overlap with those of other conditions, such as pyelonephritis, renal abscess, or even renal malignancy [8]. This diagnostic ambiguity frequently results in delayed recognition.
Radiographic findings are similarly nonspecific. Typically, Computed Tomography (CT) scans reveal enlarged kidneys with focal or diffuse low-attenuation lesions that may be misinterpreted as abscesses, tumors, or xanthogranulomatous pyelonephritis [9]. The presence of subcapsular collections, as observed in this case, further complicates the differential diagnosis process.
A definitive diagnosis necessitates a histopathological examination that demonstrates the characteristic triad of: The presence of von Hansemann cells, characterized by their eosinophilic cytoplasm, was observed. Additionally, Michaelis-Gutmann bodies were identified, along with positive PAS and iron staining [10-12]. While percutaneous renal biopsy is a reliable method for establishing a diagnosis, the focal nature of the lesions may result in sampling error. In cases where a definitive diagnosis is uncertain or there is a high suspicion of malignancy, surgical excision may be necessary for a definitive diagnosis, as was the case in the patient presented.
The differential diagnosis of renal malakoplakia encompasses a range of conditions that may present with analogous clinical and radiographic characteristics.
Xanthogranulomatous pyelonephritis is the most significant differential diagnosis [11]. This chronic inflammatory condition manifests similarly to recurrent urinary tract infections and may demonstrate analogous imaging findings. Histologically, both conditions feature lipid-laden macrophages; however, xanthogranulomatous pyelonephritis is distinguished by the absence of Michaelis-Gutmann bodies. It is noteworthy that these two conditions have been documented to coexist within the same patient, as reported in the extant literature.
It is imperative to rule out renal cell carcinoma, particularly in cases that present with renal masses. The imaging characteristics of these conditions may overlap, and a definitive diagnosis often requires histopathological examination.
Renal abscess can present with similar clinical features and imaging findings. However, abscesses generally exhibit more pronounced rim enhancement and may be associated with gas formation in cases of gas-forming organisms.
In endemic regions or among immunocompromised individuals, the consideration of tuberculous pyelonephritis is particularly relevant. The presence of caseating granulomas on histological examination serves as a distinguishing characteristic between tuberculosis and malakoplakia.
In our patient, several specific findings helped navigate the complex differential diagnosis:
* Xanthogranulomatous Pyelonephritis (XGP): While the CT scan showing a multiloculated mass and the presence of calculi initially suggested XGP, the definitive absence of Michaelis-Gutmann bodies in the surgical specimen ruled this out.
* Renal Cell Carcinoma (RCC): The rapid clinical onset and the purulent nature of the collection drained percutaneously were less typical for RCC, although the renal mass appearance required histopathological exclusion.
* Tuberculous Pyelonephritis: This was explicitly ruled out by negative specialized testing for Mycobacterium tuberculosis and the absence of caseating granulomas on histology.
The optimal management of renal malakoplakia remains controversial due to the rarity of the condition and the paucity of randomized controlled trials. Treatment strategies encompass antimicrobial therapy, immunomodulation, and surgical intervention.
Antimicrobial therapy is the foundation of medical management. Agents demonstrating effective intracellular penetration are preferable, as they are capable of reaching bacteria sequestered within macrophages [2,8]. Fluoroquinolones (ciprofloxacin, levofloxacin) and trimethoprim-sulfamethoxazole have demonstrated efficacy in multiple case reports and series. In recent studies, azithromycin has demonstrated significant potential due to its substantial intracellular accumulation and its effectiveness against Gram-negative organisms. The duration of treatment typically ranges from several weeks to months, with some cases necessitating prolonged suppressive therapy. In this case, the isolated multidrug-resistant E. coli required targeted therapy with intravenous cefepime. While cefepime was selected based on the identified multidrug-resistant E. coli sensitivity, its role in the clinical outcome should be framed within broader literature-based considerations rather than as a definitive cause of treatment failure. Although cefepime effectively addressed the systemic inflammatory response, its relatively lower intracellular penetration compared to fluoroquinolones may have limited the eradication of bacteria sequestered within macrophages. However, other clinical factors likely contributed to the progression, including the severity of the initial parenchymal compression by the subcapsular collection and the patient’s underlying diabetic microangiopathy, which might have compromised local drug delivery and tissue recovery. This pharmacological limitation, combined with the 85% loss of renal function shown on the DMSA scan, reinforced the necessity of nephrectomy to remove the persistent inflammatory focus.
Adjunctive immunomodulatory therapies have been employed in select cases. The administration of cholinergic agonists, such as bethanechol, has been proposed as a method to enhance intracellular cGMP levels, thereby theoretically improving the functionality of macrophages. Ascorbic acid (vitamin C) has also been proposed to enhance bacterial killing. Nevertheless, the extant evidence supporting these interventions remains limited to case reports. In our case, adjunctive immunomodulatory therapies were not pursued, given the severe and irreversible parenchymal destruction. The 85% loss of differential renal function rendered conservative medical optimization futile, making surgical clearance the only viable definitive treatment.
Surgical intervention, ranging from percutaneous drainage to nephrectomy, may be necessary in several scenarios, including the following: failure of medical therapy, extensive parenchymal destruction with a non-functional kidney, diagnostic uncertainty with concern for malignancy, or complications such as abscess formation or hemorrhage [9].
In this particular instance, the decision to proceed with nephrectomy was multifactorial. Despite an initial clinical improvement with drainage and antibiotics, functional imaging revealed severe irreversible parenchymal damage with only 15% differential function. The patient’s non-functional renal status, in conjunction with the potential for recurrent infection and the diagnostic uncertainty that preceded histopathological confirmation, served as the rationale for the surgical excision procedure. This approach aligns with the findings in the extant literature, suggesting that nephrectomy may be an appropriate treatment option for unilateral disease with a non-functional kidney, particularly in cases where medical therapy has failed to achieve resolution [9-11].
Historically, renal parenchymal malakoplakia has been associated with a poor prognosis, with mortality rates frequently exceeding 50% [2]. However, contemporary series report significantly improved outcomes, likely reflecting earlier diagnosis, effective antimicrobial therapy, and better supportive care [8]. In many cases, patients treated with appropriate antibiotics, particularly fluoroquinolones, demonstrate favorable outcomes with preservation of renal function.
The pattern of renal involvement has been demonstrated to influence prognosis. Unifocal disease is generally associated with a more favorable prognosis in comparison to multifocal or bilateral involvement. Early diagnosis and treatment initiation before extensive parenchymal destruction occurs are critical for renal preservation.
For patients requiring nephrectomy, long-term outcomes are generally favorable provided the contralateral kidney functions normally [9,11,12]. However, these patients require ongoing nephrological surveillance, particularly those with underlying conditions such as diabetes that increase the risk for chronic kidney disease progression.
The patient’s positive 3-month outcome, characterized by complete symptom resolution and stable renal function, is encouraging. Ongoing surveillance must be maintained in order to monitor for the potential occurrence of complications and to optimize the management of the patient’s solitary kidney.
This case contributes to the literature by illustrating a paradoxical clinical trajectory: a successful clinical and biological recovery following drainage and targeted antibiotics, which masked a complete and irreversible functional loss of the kidney. Unlike many reported cases where treatment response is judged by symptom resolution, our findings emphasize the critical role of early functional imaging (DMSA scan) in revealing underlying parenchymal destruction that persists despite clinical improvement.
Renal malakoplakia is a rare but significant etiology of renal parenchymal disease, particularly in immunocompromised patients with recurrent urinary tract infections. This case underscores several pivotal clinical lessons. Firstly, malakoplakia should be considered in the differential diagnosis of renal masses or collections in patients with diabetes mellitus and recurrent E. coli urinary tract infections. A definitive diagnosis necessitates histopathological evidence of Michaelis-Gutmann bodies, as substantiated by characteristic Periodic Acid Schiff (PAS) staining and iron staining. Early diagnosis and prolonged antibiotic therapy with intracellular-penetrating agents may preserve renal function; however, nephrectomy remains necessary when irreversible parenchymal damage has occurred. Patients with solitary kidneys after nephrectomy for malakoplakia require continuous nephrological surveillance and optimization of underlying conditions, such as diabetes mellitus. The optimal management of complex cases is contingent upon multidisciplinary team discussion. As diagnostic awareness increases and antimicrobial therapy improves, the prognosis for renal malakoplakia continues to improve. Nevertheless, the primary focus should remain on the prevention of such complications through the effective management of underlying health conditions and the timely treatment of urinary tract infections.
Declaration
Ethical approval: Ethics approval has been obtained to proceed with the current study. Ethical approval for this study (Ethical Committee N009-24) was provided by the Ethical Committee of Ibn University Hospitals, Rabat, Morocco, on 22 January 2024.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of the journal.
Guarantor of submission: The corresponding author is the guarantor of the submission.
Availability of data and materials: Supporting material is available if further analysis is needed.
- Chaudhry AP, Satchidanand SK, Anthone R, Baumler RA, Gaeta JF. An unusual case of supraclavicular and colonic malakoplakia--a light and ultrastructural study. J Pathol. 1980;131(3):193-208. Available from: https://pubmed.ncbi.nlm.nih.gov/6253612/
- Dobyan DC, Truong LD, Eknoyan G. Renal malacoplakia reappraised. Am J Kidney Dis. 1993;22(2):243-52. Available from: https://pubmed.ncbi.nlm.nih.gov/8352248/
- Mitchell MA, Markovitz DM, Killen PD, Braun DK. Bilateral renal parenchymal malacoplakia presenting as fever of unknown origin: case report and review. Clin Infect Dis. 1994;18(5):704-18. Available from: https://pubmed.ncbi.nlm.nih.gov/7915547/
- Zhang Y, Byrnes K, Lam-Himlin D, Pittman M, Pezhouh M, Gonzalez RS, et al. Gastrointestinal malakoplakia: clinicopathologic analysis of 26 cases. Am J Surg Pathol. 2020;44(9):1251-1258. Available from: https://pubmed.ncbi.nlm.nih.gov/32301754/
- Teeters JC, Betts R, Ryan C, Huether J, Elias K, Hartmann D, et al. Rectal and cutaneous malakoplakia in an orthotopic cardiac transplant recipient. J Heart Lung Transplant. 2007;26(4):411-3. Available from: https://pubmed.ncbi.nlm.nih.gov/17403486/
- Chou SC, Wang JS, Tseng HH. Malacoplakia of the ovary, fallopian tube, and uterus: a case associated with diabetes mellitus. Pathol Int. 2002;52(12):789-93. Available from: https://pubmed.ncbi.nlm.nih.gov/12588449/
- Chen CS, Lai MK, Hsueh S, Hwang TL, Chuang CK. Renal malacoplakia with secondary hepato-duodenal involvement. J Urol. 1994;151(4):982-5. Available from: https://pubmed.ncbi.nlm.nih.gov/8126844/
- Tam VK, Kung WH, Li R, Chan KW. Renal parenchymal malacoplakia: a rare cause of ARF with a review of recent literature. Am J Kidney Dis. 2003;41(6):E13-E17. Available from: https://pubmed.ncbi.nlm.nih.gov/12776305/
- Fatola A, Johnson BC, Walsh L, Fang D, White MJ, Le D, et al. A very complicated UTI: malakoplakia following E. coli urinary tract infection. BMC Nephrol. 2024;25(1):200. Available from: https://pubmed.ncbi.nlm.nih.gov/38890600/
- Chaudhry N, Vazzano J, Parwani A. Case study: malakoplakia of the bladder. Pathol Res Pract. 2022;237:153852. Available from: https://pubmed.ncbi.nlm.nih.gov/35878530/
- Pan Y, Hong YC, Shih HJ, Chang C, Huang S, Wu S, et al. Malakoplakia and xanthogranulomatous pyelonephritis treated with nephrectomy: a case report. Medicine (Baltimore). 2021;100(36):e27137. Available from: https://pubmed.ncbi.nlm.nih.gov/34516505/
- Wang Z, Ren J. Clinical analysis of renal failure caused by malakoplakia: a case report and literature review. Front Med (Lausanne). 2022;9:770731. Available from: https://www.researchgate.net/publication/358987906_Clinical_Analysis_of_Renal_Failure_Caused_by_Malakoplakia_A_Case_Report_and_Literature_Review